Shipping Temperature-Sensitive Materials: Considerations in Packaging Selection
By: Berlin Packaging Specialist
Date: October 28, 2019
Lives depend on the safe and secure transport of vaccines and pharmaceuticals as well as biological substances such as plasma, blood, organs, and tissue. The market for these types of products is enormous. The biopharmaceutical sector alone contributes more than $917 billion to the U.S. economy annually.1
Many of these critical products are sensitive to temperature and need to be shipped in a controlled, “cold chain” supply system. All of this adds complexity and, combined with the value and importance of the products, risk mitigation is essential. Two of the greatest risks in the supply chains of temperature-sensitive products are adulteration due to damage or temperature excursions (deviation from required thermal ranges) and non-compliance with applicable laws and regulations. The costs for temperature excursions are high. Another study found that by cutting shipping excursions and site temperature deviations in half, a small company with 150 clinical trials worldwide could save over $2 million and free up at least eight people to perform other work.2
Both of these risks can be mitigated with the right supply-chain expertise. In this paper, we focus on understanding the cold chain and temperature-controlled packaging. We will address:
- Cold chain basics
- Temperature-controlled packaging basics
- Packaging selection: materials
- Packaging selection: variables
- Useful resources
Before discussing packaging, we will begin with the basics of a cold chain system.
Cold Chain Basics
The “cold chain” is a logistics network designed to maintain optimal conditions during the packaging, handling, labeling, shipping, and storage of perishable items. The goal is to ensure that perishable products arrive at the end-user in a usable state. This means that the cold chain system must maintain a temperature range specific for the products being shipped.
Temperature excursions cannot be tolerated, but transit risks can be great. Airport tarmac temperatures can climb quickly during staging and loading, yet at a typical cruising altitude for an aircraft, temperatures can plummet to -20 °C. Natural disasters, poor weather, or human-caused events such as delays at customs can also interrupt the planned transport route and unexpectedly increase time in transit.
Temperature-Controlled Packaging Overview
Temperature-controlled packaging (TCP) is one element of the cold chain. TCP is designed and validated to rigorous standards to keep products within a specific temperature range for a given time period. Many pharmaceuticals, for example, need to stay between 2 °C and 8 °C. Other materials need to stay frozen; still others must maintain a controlled room-temperature state, such as 15 °C to 25 °C.
There are three types of TCP solutions: active, passive, and hybrid systems. Active systems include refrigerated boxes and pallet shippers, which operate via a power source like a lithium battery. Passive systems rely on insulated packaging along with a refrigerant, such as wet or dry ice, gel packs or wraps, foam bricks, phase change materials (PCM), or liquid nitrogen. And hybrid TCP systems incorporate elements from both active and passive systems.
Passive systems are practical, cost-effective, and are able to be designed to suit many situations. We will focus the remainder of this paper on passive solutions. 
A common passive TCP shipper consists of:
- An outer corrugated carton
- An inner cooler
- Refrigerant
- Primary containers (such as specimen bags or vials)
- Protective packaging (such as peanuts or “dunnage” to fill void spaces)
- A leak-proof bag and absorbent (for air transport of Category A and B infectious or biological substances)
The system is elaborate to ensure the required temperature parameters are met. Indeed, for many pharmaceuticals, medical devices, biotechnology products, and chemicals, the packaging system must be tested and proven to meet performance standards required by various regulatory agencies. These include the U.S. Food and Drug Administration (FDA), the U.S. Department of Transportation (DOT), and the International Air Transport Association (IATA). (Although IATA is not a regulatory body, the majority of the world’s air carriers specify the rules under which they will transport hazardous materials in IATA’s Dangerous Goods Regulations.)
Some of the standard tests performed to validate the packaging system’s ability to maintain temperature include the International Safe Transit Association (ISTA) standards 3A, 20, 7D, and 7E as well as the ASTM International standard D3103.
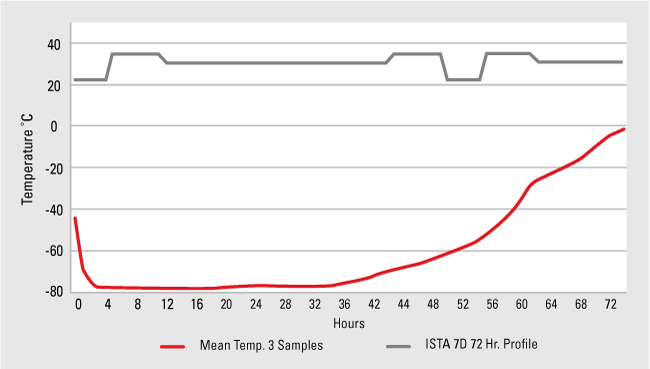
For example, ISTA has written the 7D profile to include both summer and winter conditions, since hot and cold extremes can vary depending on time of year. The profile defines durations of specific temperature ranges the packaging system must handle; these ranges are programmed into a special thermal chamber to test the package. The graph shown below shows the results for an ISTA 7D summer profile test, showing that the given packaging system (in this case an EPS foam thermal shipper) will stay frozen (below 0 °C) for up to 72 hours. The gray line is the programmed temperature profile used within the thermal chamber; the red line is the mean temperature of three sample packaging systems tested within this chamber. In this test, the samples met the standard by staying below 0 °C for the required time.
ISTA 7D Results
TCP can also be subjected to the rigorous test procedures specified in Title 49 of the Code of Federal Regulations (CFR). This ensures hazardous temperature-sensitive materials such as aerospace adhesives (flammable liquids/solids), infectious substances, and radio-pharmaceuticals (radioactive) can meet the performance test requirements for the transport of dangerous goods. The 49 CFR tests subject the packaging system to shocks, drops, vibration, water, and pressure in order to achieve a United Nations (UN) rating. Going into detail here is beyond the scope of this paper, but the Useful Resources section of this document provides suggestions for learning more.
Packaging Selection: Materials
A passive TCP system includes an insulated package and a refrigerant. To best control the temperature and protect against physical damage during shipment, there are a number of different factors to consider.
The product being shipped:
- What are the physical characteristics of the material to be shipped?
- What temperature range needs to be maintained and for how long?
- What regulations impact packaging selection?
The transit corridor:
- What transit conditions (vibration, shocks, extreme temperatures, etc.) do you expect?
- What season(s) do you expect to ship in?
- What temperature ranges will the shipment be exposed to during transit?
- How long will the package be in transit?
Packaging use and cost:
- Do you plan to re-use the packaging? Can it be re-used? How many times?
- Based on the value of the payload and the cost of an excursion, what is the total cost you’re willing to absorb?
Insulating Materials
The three most common types of insulated packaging for non-bulk shipments of temperature-sensitive materials are expanded polystyrene (EPS) foam, polyurethane (PUR) foam, and vacuum insulated panels (VIP). The relative merits of each type are detailed in the table below.
| Expanded Polystrene (EPS) Foam | Polyurethane (PUR) Foam | Vacuum Insulated Panels (VIPs) | |
|---|---|---|---|
| Definition | A closed-cell, lightweight, rigid-foam plastic typically molded (rather than formed) into panels. | A rigid piece of polyurethane foam molded into shape or used as an insulating panel. | A special insulating material (e.g., carbon) formed into a solid core and then vacuum-sealed. |
| Benefits | • Lightweight • Inexpensive • Recyclable • Re-usable • Superior dry ice sublimation | • Sturdy protection from rough distribution environments • Re-usable many times | Lower dimensional weight reduces shipping costs • Requires less refrigerant • Recyclable • Re-usable many times |
| Trade-Offs | • Doesn’t withstand multiple re-use as well as PUR or VIP • Bulkier design requires lots of storage space | • More expensive than EPS • Cannot be recycled | • More expensive than EPS or PUR • Can lose their vacuum seal, decreasing efficiency |
| R-Value3 | • Lowest | • Medium | • Highest |
| Best Suited For | • Refrigerated or frozen materials shipped within a short transit duration | • Larger payloads • Extended domestic shipping windows | • Highly temperature-sensitive shipments • High-value shipments • Extended shipping windows • International shipments |
In addition to the insulating materials detailed above, the cold chain marketplace offers a myriad of other options to explore, including insulated pallet shippers, insulating (soft-sided) pouches, hard-cased thermal chests and boxes, cryogenic units, and active thermal systems.
Refrigerants
Three of the more common refrigerants are listed below along with some of the relative merits of each type. Keep in mind that refrigerants and insulating packaging must be considered as a complete system.
| Dry Ice | Gel Packs | Phase Change Material (PCM) | |
|---|---|---|---|
| Definition | The solid form (typically in pellets or blocks) of carbon dioxide. | A rigid plastic container or soft-sided pouch containing a liquid (e.g., water or refrigerant gel) available in a variety of shapes, sizes, and thicknesses. | A material that changes phases (from solid to liquid or liquid to gas) at a given temperature. |
| Benefits | • Inexpensive • Readily available • Does not require pre-conditioning | • Can be re-washed, re-conditioned, and then re-used • Simplifies pack-outs; no need to weigh it as the packaging system s Other refrigerants used in the cold chain include foam bricks, gel mats, wet ice, thermal blankets, and liquid nitrogen. With an understanding of insulating materials and refrigerants, the shipper can properly evaluate specific shipping systems. For example, for a product that is highly temperature-sensitive and needs to withstand international travel conditions and potential delays, the shipper might select phase change material as the refrigerant, a cooler with a higher R-value such as polyurethane or VIP, and strong protective packaging. Third party labs and packaging partners can assist in qualifying and validating packaging to ensure it performs as intended. Packaging Selection: VariablesThere are other factors to consider when evaluating a temperature-sensitive packaging solution. System Cost
Pack-Out Simplicity
Thermal Performance
Physical Performance
Spotlight on Berlin Dangerous GoodsBerlin Packaging is a leading supplier of rigid packaging, and its Dangerous Goods division specializes in shipping systems for the safe transport of hazardous and temperature-sensitive materials. For over 30 years, the Dangerous Goods team has been designing, testing, manufacturing, and assembling UN-rated and temperature-sensitive packaging in a wide variety of styles. The division offers a selection of over 1,600 stock packaging systems as well as the ability to custom-engineer and manufacture solutions to meet a customer’s exact needs. In addition to in-house UN and thermal testing capabilities, the division uses third-party labs to independently certify UN-rated packaging systems as well as to validate thermal performance. Berlin Packaging’s Dangerous Goods division is ISO 9001:2008 certified and has a dedicated team of consultants and engineers with expert knowledge of the regulations and requirements related to shipping hazardous and temperature-sensitive materials. Getting StartedBuilding a successful cold chain transportation system requires proper packaging, proper transport and storage equipment, highly trained personnel, and efficient management procedures. Below are some tips for getting started in selecting the proper packaging. Understand the Characteristics of the MaterialWhat are the physical characteristics of the material or product to be shipped? Fragile materials will require more protective packaging; heavy materials may not be suited to EPS foam coolers unless a sturdy outer carton is also utilized. Temperature requirements and the length of time the material must be kept in a temperature range are additional factors to consider. For instance, some materials cannot tolerate being frozen so dry ice is not an option, but gel packs or phase change materials may work. Other materials are extremely sensitive to temperature fluctuations, so phase change materials might be the better option. Assess the Likely Transit CorridorsWhat is the distribution environment your material will experience? Multi-modal transport; rough road, air, or sea conditions; temperature variations; and the distance the material must traverse all affect the type of packaging system needed. For example, shipping materials during the summer from a northern U.S. city to a southern U.S. city will probably require a different packaging system or pack-out than shipping from the same point of origination to Canada because of the closer proximity and similar climate. International shipments must consider additional variables such as carrier stowage and customs clearance as well as the longer duration of the trip. Evaluate Packaging Options and CostsWhat is the total “landed” cost of the packaging, including freight and packaging costs? Optimized packaging design can reduce the total cost in several ways. Designing the package to minimize size and weight, without impacting the thermal capability, can reduce freight costs. Consolidation of packaging materials to be used frequently within different package designs can drive lower costs. Re-using packaging, or implementing a rebuild/closed-loop reverse logistics system, can also cut costs. And total costs can also consider potential product loss due to inferior packaging components (so a more expensive packaging system may actually be less expensive when product loss is factored in). Understand the RegulationsReview the regulatory requirements related to your material. If shipping in the U.S., does the 49 CFR (or the IATA Dangerous Goods Regulations) specify limits on the type or amount of material to be shipped by that mode of transportation? If you are shipping biological material, pharmaceuticals, or medical devices, are other regulatory bodies concerned with how these materials need to be shipped? The U.S. Department of Transportation, IATA, and Food and Drug Administration have helpful resources online. Becoming knowledgeable in the laws of all of the countries in your distribution channel is critical. This can help to avoid costly fines or delivery delays. Given the complexity and importance of establishing the right cold chain system, many shippers work with experts to establish and maintain the right systems. Seasoned packaging consultants are a valuable resource to locate the packaging that is appropriate for your needs. SummaryThe economic and human value of products shipped throughout the cold chain is enormous. At the same time, transit risks and regulatory rules create an environment that has to be carefully managed. Packaging plays an important role in avoiding expensive and sometimes catastrophic loss of product due to temperature excursions and/or damage to the packaging system. This paper covered key elements of passive temperature-controlled packaging systems – from the materials to the refrigerants to the variables to consider when picking a solution. By systematically evaluating the products to be shipped, the shipping dynamics, and the regulations, you can build cost-effective packaging for your cold chain. Partners exist to help each step of the way. Companies of all sizes and sophistication often look to these experts to create, supply, and manage their packaging inventory. Useful ResourcesThere are many documents that offer information and advice on handling, storage, transport, and distribution of temperature-sensitive materials. Regulations:
A selection of recommended storage and handling practices:
1 “The U.S. Biopharmaceuticals Sector: Economic Contribution to the Nation,” Battelle Technology Partnership Practice Report, July 2011. 2 “Modeling the Clinical Trial, Calculating Company-Wide Savings and Comparisons Among Several Alternative Distribution Ideas,” Pharmaceutical Outsourcing Journal, May/June 2012. 3 R-value is the capacity of an insulating material to resist heat flow. The higher the R-value, the greater the insulating power. 4 Shippers must first follow the 49 CFR, but then consult IATA guidelines for carrier rules and exceptions. |
